Evaluating BON Design
BiodiversityObservationNetworks.jl contains several utilities for measuring properties of a BON. These properties can be split into two categories: (1) properties that measure spatial balance and (2) properties that measure environmental balance.
Let's load the packages and a couple functions from StatsBase
using BiodiversityObservationNetworks
using BiodiversityObservationNetworks.StatsBase: mean, std
using CairoMakie
using SpeciesDistributionToolkitRandom.TaskLocalRNG()There are three metrics for evaluating BON design: two measure the spatial balance of a sample (MoransI and VoronoiVariance), and one measures how representative the environmental conditions at BON are relative to the domain being sampled (``).
Moran's I
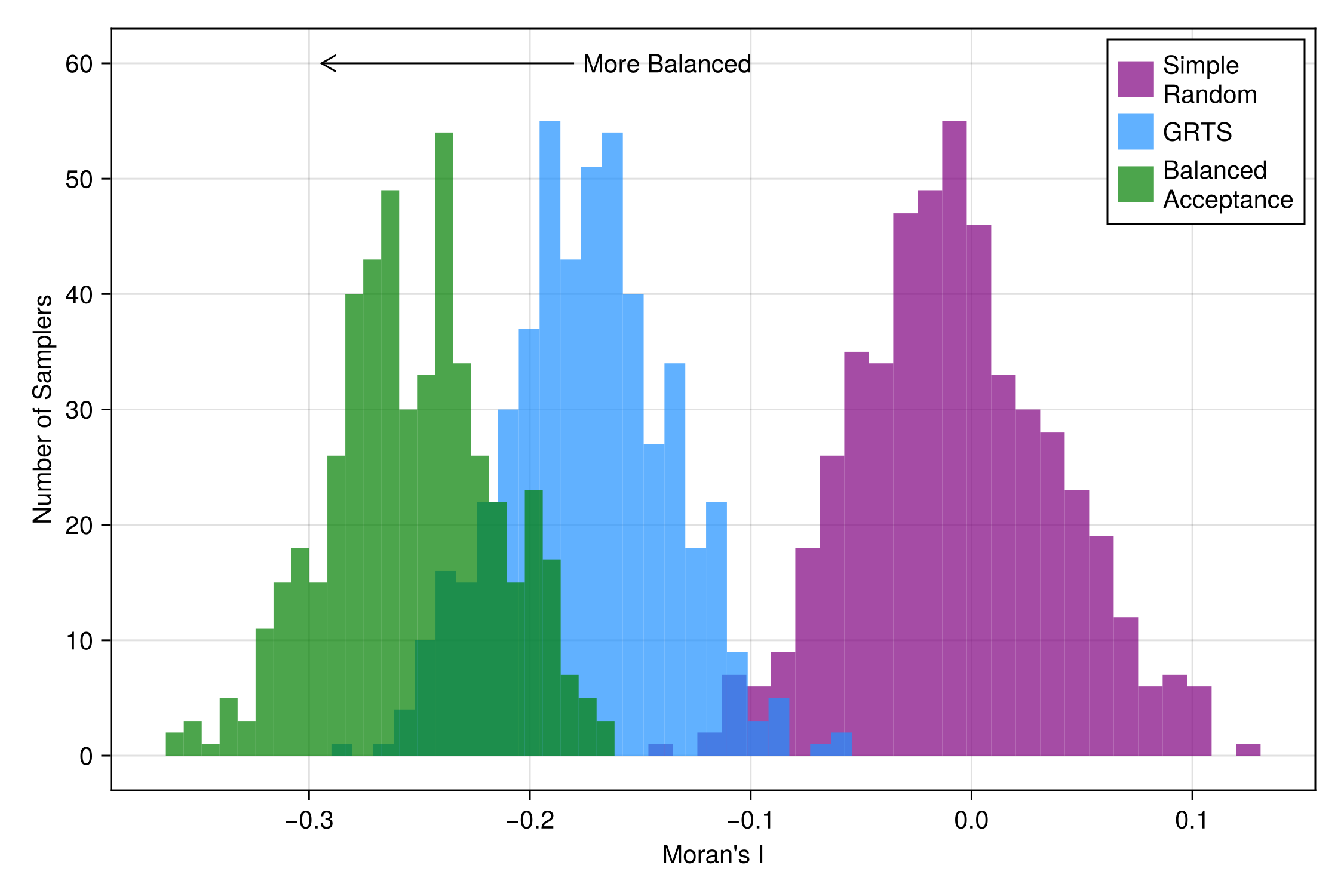
MoransI is a metric that calculates spatial autocorrelation of the sample indicator variable across the domain (1 if a site is sampled, 0 otherwise). Negative values indicate spatial inhibition (spread), which is desired for balanced sampling. This version was specifcally proposed by (Tillé et al., 2025).
For these examples, we'll use a 30 x 30 matrix of zeros as our domain, but any domain will work here
domain = zeros(30, 30);Let's compare spatial balance (using MoransI) for three different sampling algorithms: (1) SimpleRandom, (2) BalancedAcceptance and (3) GRTS.
We'll use 500 random BON samples per sampling method
reps = 500500And use in-line loops to record Moran's
grts_I = [spatialbalance(MoransI(), domain, sample(GRTS(), domain)) for r in 1:reps];
bas_I = [spatialbalance(MoransI(), domain, sample(BalancedAcceptance(), domain)) for r in 1:reps];
srs_I = [spatialbalance(MoransI(), domain, sample(SimpleRandom(), domain)) for r in 1:reps];And visualize the scores of each sampler.
Code for the figure
f = Figure(size=(750,500))
NUM_BINS = 25
ax = Axis(f[1,1], xlabel = "Moran's I", ylabel = "Number of Samplers")
hist!(ax, srs_I, color=(:purple, 0.7), bins=NUM_BINS, label = "Simple\nRandom")
hist!(ax, grts_I, color=(:dodgerblue, 0.7), bins=NUM_BINS, label = "GRTS")
hist!(ax, bas_I, color=(:green, 0.7), bins=NUM_BINS, label = "Balanced\nAcceptance")
annotation!(ax, 200, 0, -0.3, 60,
text = "More Balanced",
style = Ann.Styles.LineArrow()
)
axislegend(position = :rt)As we can see (and is consistent with the results in the literature that developed these samplering algorithms), GRTS is more spatially balanced than Simple Random Sampling, but is outperformed by Balanced Acceptance Sampling.
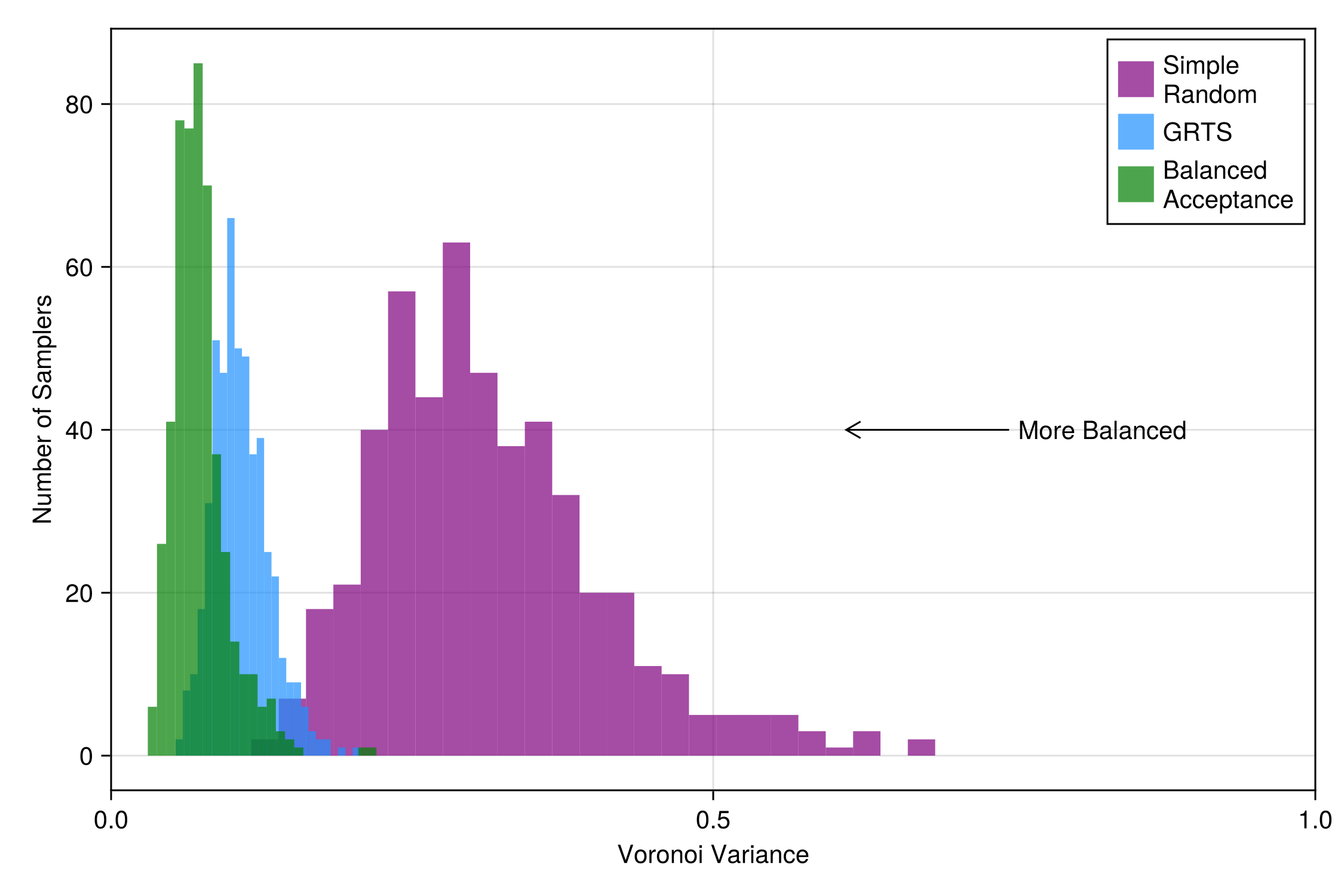
VoronoiVariance
An alternative method for evaluating spatial balance is by using the Voronoi tesselation, which splits the domain into a series of polygons, where the bon is the
These polygons can then be used to assess the spatial balance of a sample. In an ideally balanced sample, the sum of the inclusion probabilities within each polygon
If we define i, i.e.
then we can assess the spatial balance of a sample by measuring the distance of
(Grafström, 2025) proposes the metric B, defined as
to measure spatial balance, where smaller values indicate better spatial balance.
We'll now compare VoronoiVariance across samplers in the same way as above:
grts_vv = [spatialbalance(VoronoiVariance(), domain, sample(GRTS(), domain)) for r in 1:reps];
bas_vv = [spatialbalance(VoronoiVariance(), domain, sample(BalancedAcceptance(), domain)) for r in 1:reps];
srs_vv = [spatialbalance(VoronoiVariance(), domain, sample(SimpleRandom(), domain)) for r in 1:reps];and visualize
Code for the figure
f = Figure(size=(750,500))
NUM_BINS = 25
ax = Axis(f[1,1], xlabel = "Voronoi Variance", ylabel = "Number of Samplers")
hist!(ax, srs_vv, color=(:purple, 0.7), bins=NUM_BINS, label = "Simple\nRandom")
hist!(ax, grts_vv, color=(:dodgerblue, 0.7), bins=NUM_BINS, label = "GRTS")
hist!(ax, bas_vv, color=(:green, 0.7), bins=NUM_BINS, label = "Balanced\nAcceptance")
annotation!(ax, 150, 0, 0.6, 40,
text = "More Balanced",
style = Ann.Styles.LineArrow()
)
xlims!(ax, 0,1)
axislegend(position = :rt)and again we see that GRTS is more spatially balanced than Simple Random Sampling, but is outperformed by Balanced Acceptance Sampling.
JensenShannon
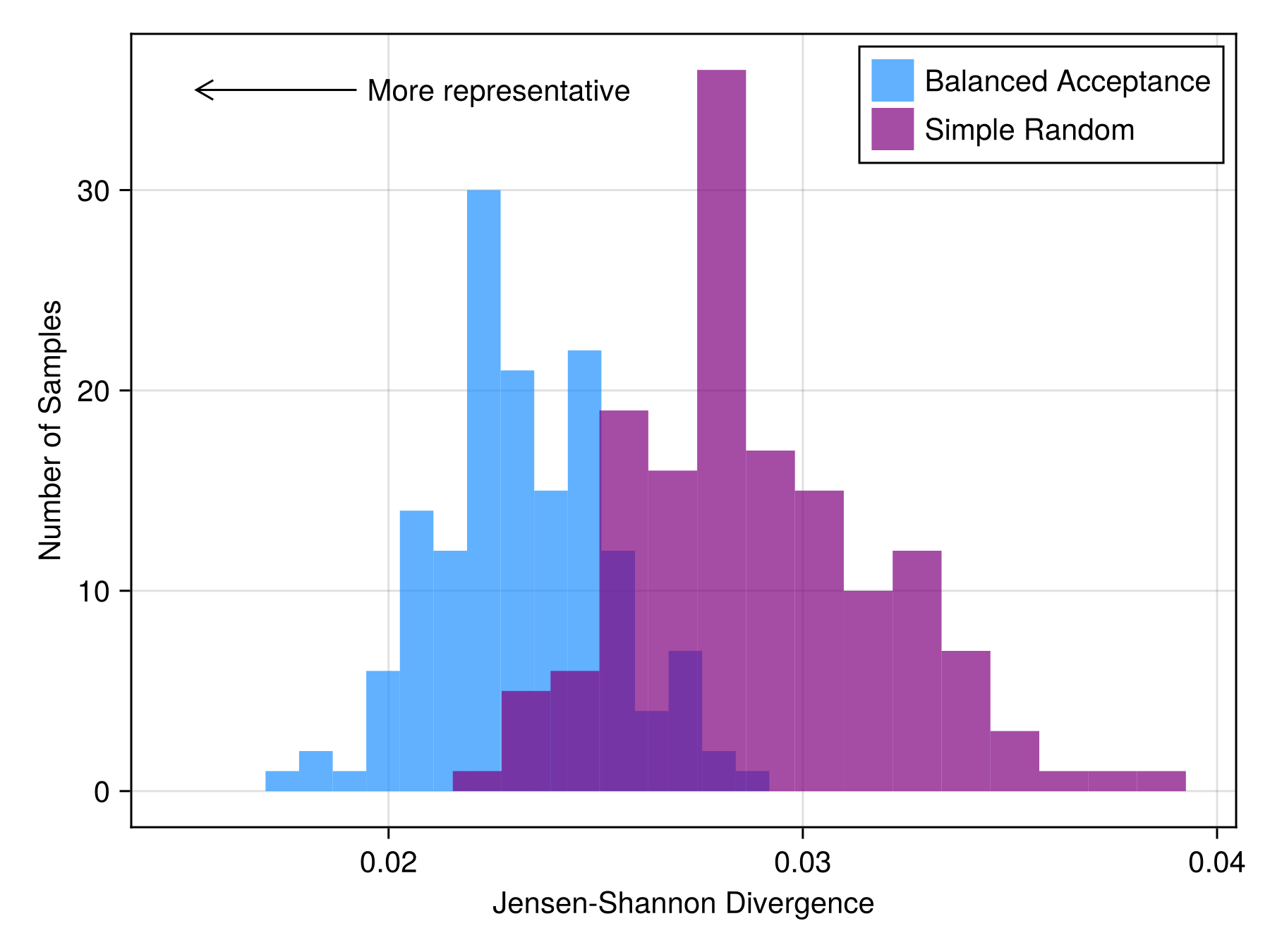
The final evaluation metric is for computing how representative a BiodiversityObservationNetwork is of the environmental variables across the domain using Jensen-Shannon divergence, a method for measuring the distance between two distributions.
Here we will use bioclimatic variables in Washington State as an example
aoi = getpolygon(PolygonData(OpenStreetMap, Places), place = "Washington State")FeatureCollection with 1 features, each with 10 propertiesWe can then load the 19 Bioclimatic variables from CHELSA as follows.
bioclim = [SDMLayer(RasterData(CHELSA1, BioClim); SpeciesDistributionToolkit.SimpleSDMPolygons.boundingbox(aoi)..., layer = i) for i in 1:19]19-element Vector{SimpleSDMLayers.SDMLayer{Int16}}:
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)
🗺️ A 416 × 952 layer (355723 Int16 cells)And mask them to just Washington state.
mask!(bioclim, aoi)19-element Vector{SimpleSDMLayers.SDMLayer{Int16}}:
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)
🗺️ A 416 × 952 layer (303225 Int16 cells)The API for [JensenShannon] is similar to other evaluation utilties:
bon = sample(SimpleRandom(), bioclim)
js = evaluate(JensenShannon(), bioclim, bon)0.05318806815211255We can then compare BalancedAcceptance to SimpleRandom:
nreps = 150
bas = [evaluate(JensenShannon(), bioclim, sample(BalancedAcceptance(100), bioclim)) for i in 1:nreps];
srs = [evaluate(JensenShannon(), bioclim, sample(SimpleRandom(100), bioclim)) for i in 1:nreps];and visualize
Code for the figure
f = Figure()
ax = Axis(f[1,1], xlabel = "Jensen-Shannon Divergence", ylabel = "Number of Samples")
hist!(bas, color=(:dodgerblue, 0.7), label = "Balanced Acceptance")
hist!(srs, color=(:purple, 0.7), label="Simple Random")
annotation!(ax, 150, 0, 0.015, 35,
text = "More representative",
style = Ann.Styles.LineArrow()
)
axislegend(position = :rt)Note that the spatially balanced sampler, BalancedAcceptance, is more environmentally representative than SimpleRandom.